SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.
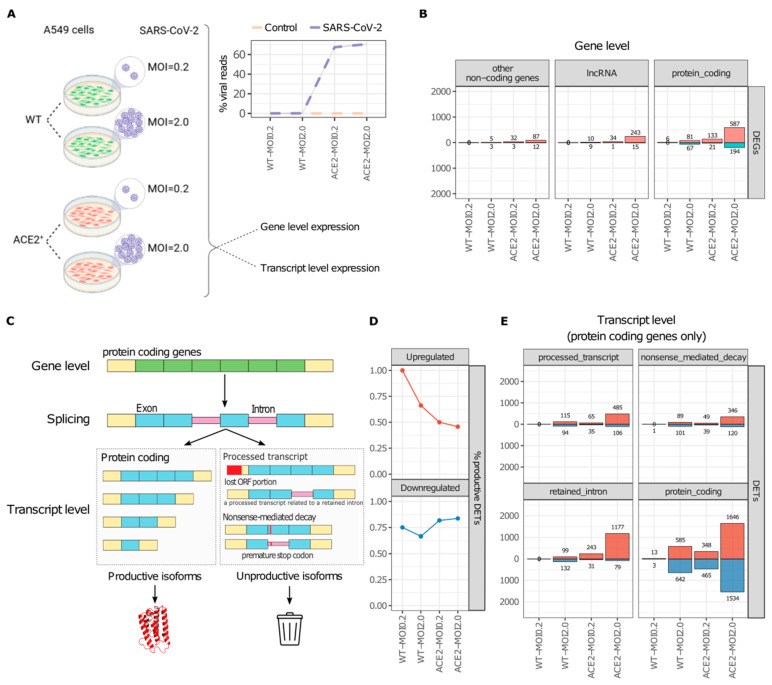
RNA processing is a highly conserved mechanism that serves as a pivotal regulator of gene expression. Alternative processing generates transcripts that can still be translated but lead to potentially nonfunctional proteins. A plethora of respiratory viruses, including severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), strategically manipulate the host's RNA processing machinery to circumvent antiviral responses. We integrated publicly available omics datasets to systematically analyze isoform-level expression and delineate the nascent peptide landscape of SARS-CoV-2-infected human cells. Our findings explore a suggested but uncharacterized mechanism, whereby SARS-CoV-2 infection induces the predominant expression of unproductive splicing isoforms in key IFN signaling, interferon-stimulated (ISGs), class I MHC, and splicing machinery genes, including IRF7, HLA-B, and HNRNPH1. In stark contrast, cytokine and chemokine genes, such as IL6 and TNF, predominantly express productive (protein-coding) splicing isoforms in response to SARS-CoV-2 infection. We postulate that SARS-CoV-2 employs an unreported tactic of exploiting the host splicing machinery to bolster viral replication and subvert the immune response by selectively upregulating unproductive splicing isoforms from antigen presentation and antiviral response genes. Our study sheds new light on the molecular interplay between SARS-CoV-2 and the host immune system, offering a foundation for the development of novel therapeutic strategies to combat COVID-19.
Authors
Dias TL, Mamede I, de Toledo NE, Queiroz LR
External link
Publication Year
Publication Journal
Associeted Project
Long Noncoding RNAs
Lista de serviços
-
As antisense RNA gets intronic.As antisense RNA gets intronic.
-
Androgen responsive intronic non-coding RNAs.Androgen responsive intronic non-coding RNAs.
-
Conserved tissue expression signatures of intronic noncoding RNAs transcribed from human and mouse loci.Conserved tissue expression signatures of intronic noncoding RNAs transcribed from human and mouse loci.
-
The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation.The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation.
-
Antisense intronic non-coding RNA levels correlate to the degree of tumor differentiation in prostate cancer.Antisense intronic non-coding RNA levels correlate to the degree of tumor differentiation in prostate cancer.
-
Insight Into the Long Noncoding RNA and mRNA Coexpression Profile in the Human Blood Transcriptome Upon Leishmania infantum Infection.Insight Into the Long Noncoding RNA and mRNA Coexpression Profile in the Human Blood Transcriptome Upon Leishmania infantum Infection.
-
Long non-coding RNAs associated with infection and vaccine-induced immunityLong non-coding RNAs associated with infection and vaccine-induced immunity
-
Comparative transcriptomic analysis of long noncoding RNAs in Leishmania-infected human macrophagesComparative transcriptomic analysis of long noncoding RNAs in Leishmania-infected human macrophages
-
SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.