Prion protein regulates invasiveness in glioblastoma stem cells.
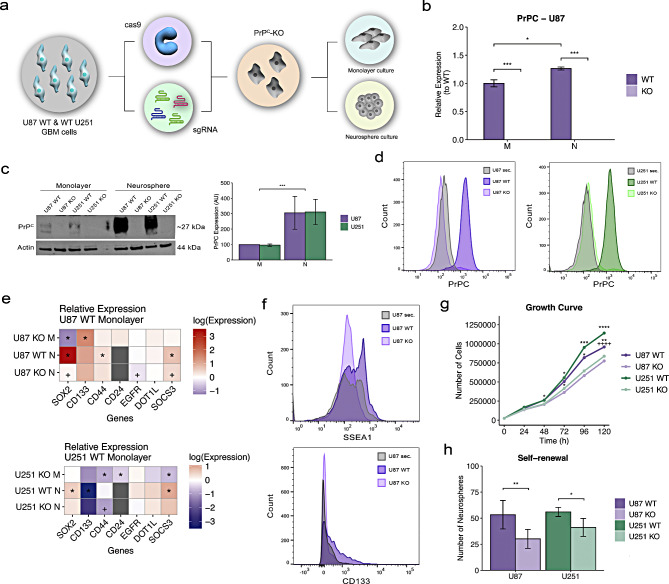
BACKGROUND: Glioblastoma (GBM) is an aggressive brain tumor driven by glioblastoma stem cells (GSCs), which represent an appealing target for therapeutic interventions. The cellular prion protein (PrPC), a scaffold protein involved in diverse cellular processes, interacts with various membrane and extracellular matrix molecules, influencing tumor biology. Herein, we investigate the impact of PrPC expression on GBM. METHODS: To address this goal, we employed CRISPR-Cas9 technology to generate PrPC knockout (KO) glioblastoma cell lines, enabling detailed loss-of-function studies. Bulk RNA sequencing followed by differentially expressed gene and pathway enrichment analyses between U87 or U251 PrPC-wild-type (WT) cells and PrPC- nockout (KO) cells were used to identify pathways regulated by PrPC. Immunofluorescence assays were used to evaluate cellular morphology and protein distribution. For assessment of protein levels, Western blot and flow cytometry assays were employed. Transwell and growth curve assays were used to determine the impact of loss-of-PrPC in GBM invasiveness and proliferation, respectively. Single-cell RNA sequencing analysis of data from patient tumors from The Cancer Genome Atlas (TCGA) and the Broad Institute of Single-Cell Data Portal were used to evaluate the correspondence between our in vitro results and patient samples. RESULTS: Transcriptome analysis of PrPC-KO GBM cell lines revealed altered expression of genes associated with crucial tumor progression pathways, including migration, proliferation, and stemness. These findings were corroborated by assays that revealed impaired invasion, migration, proliferation, and self-renewal in PrPC-KO GBM cells, highlighting its critical role in sustaining tumor growth. Notably, loss-of-PrPC disrupted the expression and localization of key stemness markers, particularly CD44. Additionally, the modulation of PrPC levels through CD44 overexpression further emphasizes their regulatory role in these processes. CONCLUSIONS: These findings establish PrPC as a modulator of essential molecules on the cell surface of GSCs, highlighting its potential as a therapeutic target for GBM.
Authors
Prado MB, Coelho BP, Iglesia RP, Alves RN, Boccacino JM
External link
Publication Year
Publication Journal
Associeted Project
Systems Immunology of Human Diseases
Lista de serviços
-
As antisense RNA gets intronic.As antisense RNA gets intronic.
-
Androgen responsive intronic non-coding RNAs.Androgen responsive intronic non-coding RNAs.
-
Conserved tissue expression signatures of intronic noncoding RNAs transcribed from human and mouse loci.Conserved tissue expression signatures of intronic noncoding RNAs transcribed from human and mouse loci.
-
The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation.The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation.
-
Antisense intronic non-coding RNA levels correlate to the degree of tumor differentiation in prostate cancer.Antisense intronic non-coding RNA levels correlate to the degree of tumor differentiation in prostate cancer.
-
Insight Into the Long Noncoding RNA and mRNA Coexpression Profile in the Human Blood Transcriptome Upon Leishmania infantum Infection.Insight Into the Long Noncoding RNA and mRNA Coexpression Profile in the Human Blood Transcriptome Upon Leishmania infantum Infection.
-
Long non-coding RNAs associated with infection and vaccine-induced immunityLong non-coding RNAs associated with infection and vaccine-induced immunity
-
Comparative transcriptomic analysis of long noncoding RNAs in Leishmania-infected human macrophagesComparative transcriptomic analysis of long noncoding RNAs in Leishmania-infected human macrophages
-
SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.