Transcriptomic insights into early mechanisms underlying post-chikungunya chronic inflammatory joint disease.
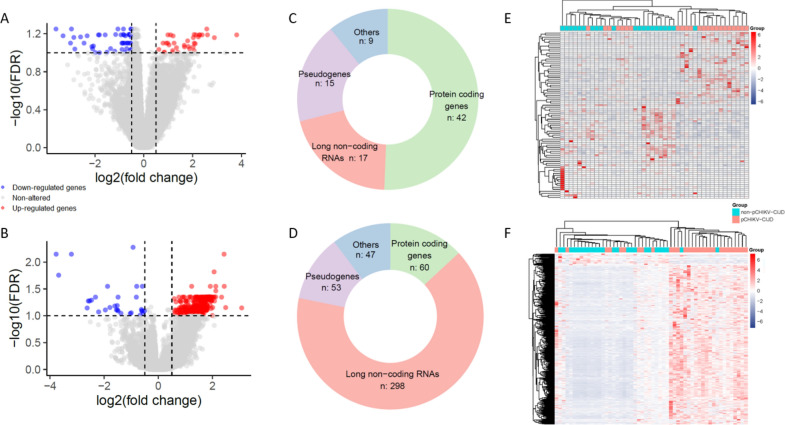
Chikungunya virus (CHIKV) infection often results in a chronic joint condition known as Post-Chikungunya Chronic Inflammatory Joint Disease (pCHIKV-CIJD). This condition disrupts individuals' daily lives and contributes to increased healthcare expenditure. This study investigated the molecular mechanisms underlying pCHIKV-CIJD development by analyzing RNA transcripts, including small RNAs, of whole blood from CHIKV-infected patients. By comparing patients who evolved to pCHIKV-CIJD with those who did not, we identified molecular signatures associated with chronification in acute and post-acute disease phases. These molecules were primarily associated with an altered immune response regulation. Notably, LIFR, an immune receptor that enhanced IL-6 transcription, was down-regulated in the acute phase of pCHIKV-CIJD patients, while its inhibitor, hsa-miR-98-5p, was up-regulated in these individuals. Other downregulated genes include members of immune mechanisms whose impairment can lead to a reduction in the first line of antiviral response, thereby promoting virus persistence for a longer period in these patients. Additionally, pCHIKV-CIJD patients exhibited reduced transcript levels of MMP8, LFT, and DDIT4, genes already implicated in the pathological process of other types of inflammatory arthritis and seemingly relevant for pCHIKV-CIJD development. Overall, our findings provide insights into the early molecular mechanisms involved in the chronification and highlight potential targets for further investigation.
Authors
Ramundo MS, da Fonseca GC, Ten-Caten F, Gerber AL, et al.
External link
Publication Year
Publication Journal
Associeted Project
Systems Immunology of Human Diseases
Lista de serviços
-
As antisense RNA gets intronic.As antisense RNA gets intronic.
-
Androgen responsive intronic non-coding RNAs.Androgen responsive intronic non-coding RNAs.
-
Conserved tissue expression signatures of intronic noncoding RNAs transcribed from human and mouse loci.Conserved tissue expression signatures of intronic noncoding RNAs transcribed from human and mouse loci.
-
The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation.The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation.
-
Antisense intronic non-coding RNA levels correlate to the degree of tumor differentiation in prostate cancer.Antisense intronic non-coding RNA levels correlate to the degree of tumor differentiation in prostate cancer.
-
Insight Into the Long Noncoding RNA and mRNA Coexpression Profile in the Human Blood Transcriptome Upon Leishmania infantum Infection.Insight Into the Long Noncoding RNA and mRNA Coexpression Profile in the Human Blood Transcriptome Upon Leishmania infantum Infection.
-
Long non-coding RNAs associated with infection and vaccine-induced immunityLong non-coding RNAs associated with infection and vaccine-induced immunity
-
Comparative transcriptomic analysis of long noncoding RNAs in Leishmania-infected human macrophagesComparative transcriptomic analysis of long noncoding RNAs in Leishmania-infected human macrophages
-
SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.