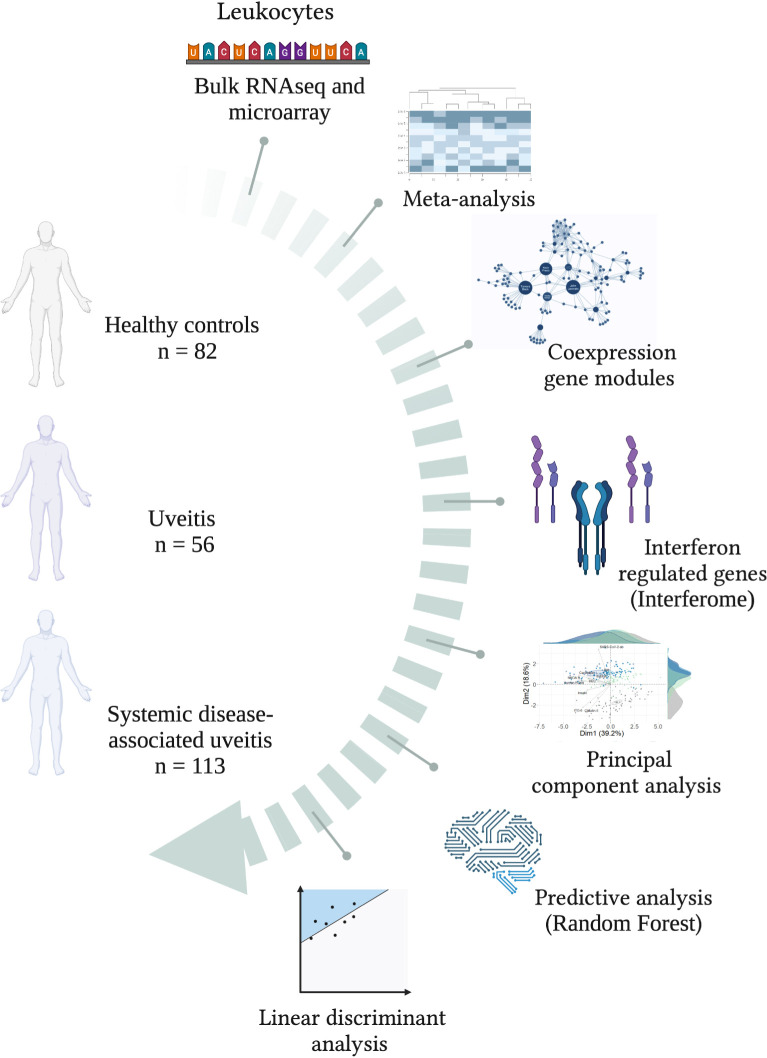
Integrative immunology identified interferome signatures in uveitis and systemic disease-associated uveitis.
INTRODUCTION: Uveitis accounts for up to 25% of global legal blindness and involves intraocular inflammation, classifed as infectious or non-infectious. Its complex pathophysiology includes dysregulated cytokines, particularly interferons (IFNs). However, the global signature of type I, II, and III interferon-regulated genes (Interferome) remains largely uncharacterized in uveitis. METHODS: In this study, we conducted an integrative systems biology analysis of blood transcriptome data from 169 non-infectious uveitis patients (56 isolated uveitis, 113 systemic disease-associated uveitis) and 82 healthy controls. RESULTS: Modular co-expression analysis identified distinct cytokine signaling networks, emphasizing interleukin and interferon pathways. A meta-analysis revealed 110 differentially expressed genes (metaDEGs) in isolated uveitis and 91 in systemic disease-associated uveitis, predominantly linked to immune responses. The Interferome database confirmed a predominance of type I and II IFN signatures in both groups. Pathway enrichment analysis highlighted inflammatory responses, including cytokine production (IL-8, IL1-β, IFN-γ, β, and α) and toll-like receptor signaling (TLR4, TLR7, TLR8, CD180). Principal component analysis emphasized the IFN signature's discriminative power, particularly in systemic disease-associated uveitis. Machine learning identified IFN-associated genes as robust predictors, while linear discriminant analysis pinpointed CCR2, CD180, GAPT, and PTGS2 as key risk factors in isolated uveitis and CA1, SIAH2, and PGS in systemic disease-associated uveitis. CONCLUSION: These findings highlight IFN-driven imune dysregulation and potential molecular targets for precision therapies in uveitis.
Authors
Munhoz DD, Fonseca DLM, Filgueiras IS, Dias HD, Nakaya HI, et al.
External link
Publication Year
Publication Journal
Associeted Project
Network & Precision Medicine
Lista de serviços
-
As antisense RNA gets intronic.As antisense RNA gets intronic.
-
Androgen responsive intronic non-coding RNAs.Androgen responsive intronic non-coding RNAs.
-
Conserved tissue expression signatures of intronic noncoding RNAs transcribed from human and mouse loci.Conserved tissue expression signatures of intronic noncoding RNAs transcribed from human and mouse loci.
-
The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation.The intronic long noncoding RNA ANRASSF1 recruits PRC2 to the RASSF1A promoter, reducing the expression of RASSF1A and increasing cell proliferation.
-
Antisense intronic non-coding RNA levels correlate to the degree of tumor differentiation in prostate cancer.Antisense intronic non-coding RNA levels correlate to the degree of tumor differentiation in prostate cancer.
-
Insight Into the Long Noncoding RNA and mRNA Coexpression Profile in the Human Blood Transcriptome Upon Leishmania infantum Infection.Insight Into the Long Noncoding RNA and mRNA Coexpression Profile in the Human Blood Transcriptome Upon Leishmania infantum Infection.
-
Long non-coding RNAs associated with infection and vaccine-induced immunityLong non-coding RNAs associated with infection and vaccine-induced immunity
-
Comparative transcriptomic analysis of long noncoding RNAs in Leishmania-infected human macrophagesComparative transcriptomic analysis of long noncoding RNAs in Leishmania-infected human macrophages
-
SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.SARS-CoV-2 Selectively Induces the Expression of Unproductive Splicing Isoforms of Interferon, Class I MHC, and Splicing Machinery Genes.